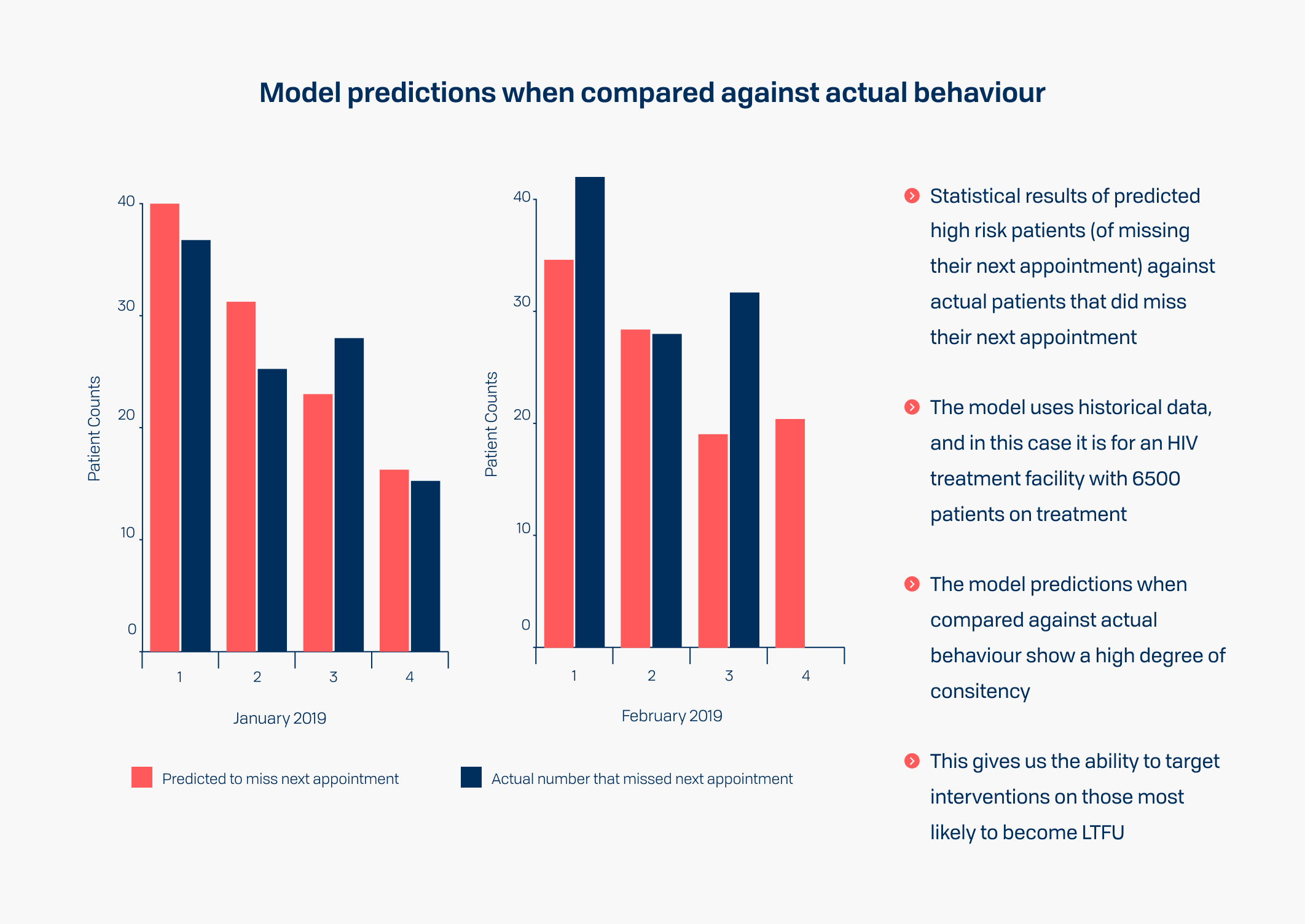
Leveraging Artificial Intelligence to produce predictive analytics that help reduce patient loss to follow up
BroadReach Consulting tackled the loss to follow up challenge by combining last mile health delivery experience with application of data science, as predictive modelling, to identify and engage patients that are most at risk of dropout as early as possible in their treatment journey.
Key benefits to the program
01. Improved patient outcomes and overall program impact
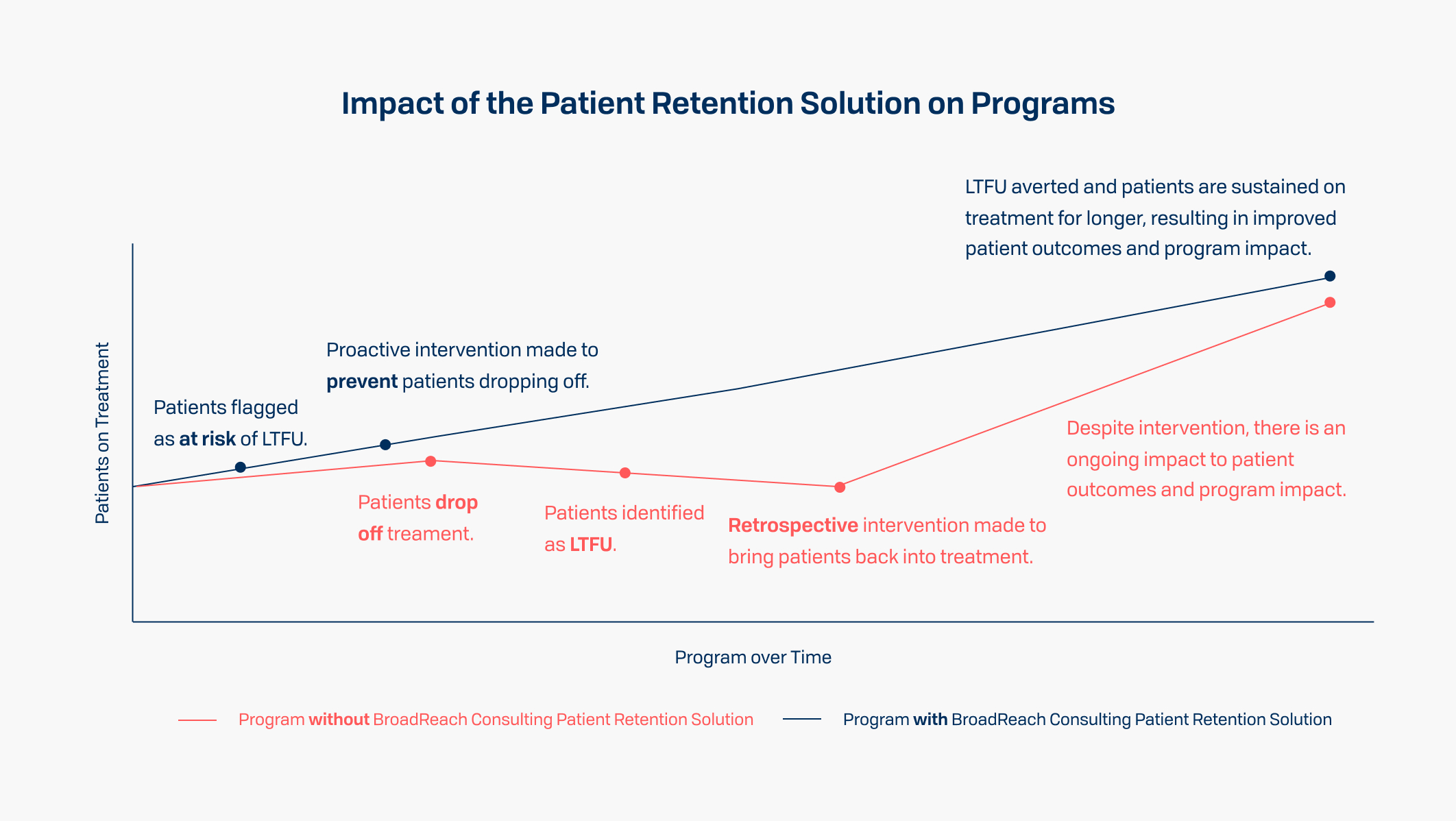
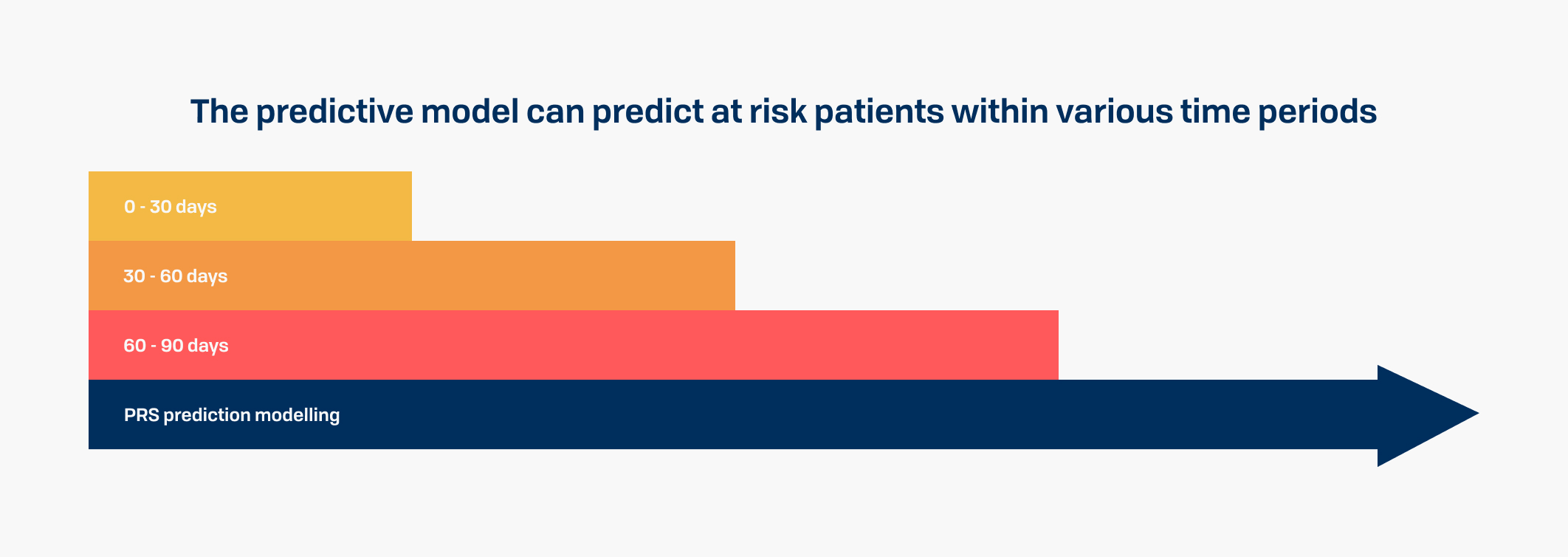
Foresight gives programs the ability to pre-emptively intervene and prevent loss to follow up, keeping more patients on treatment and saving lives.
02. Program cost efficiencies
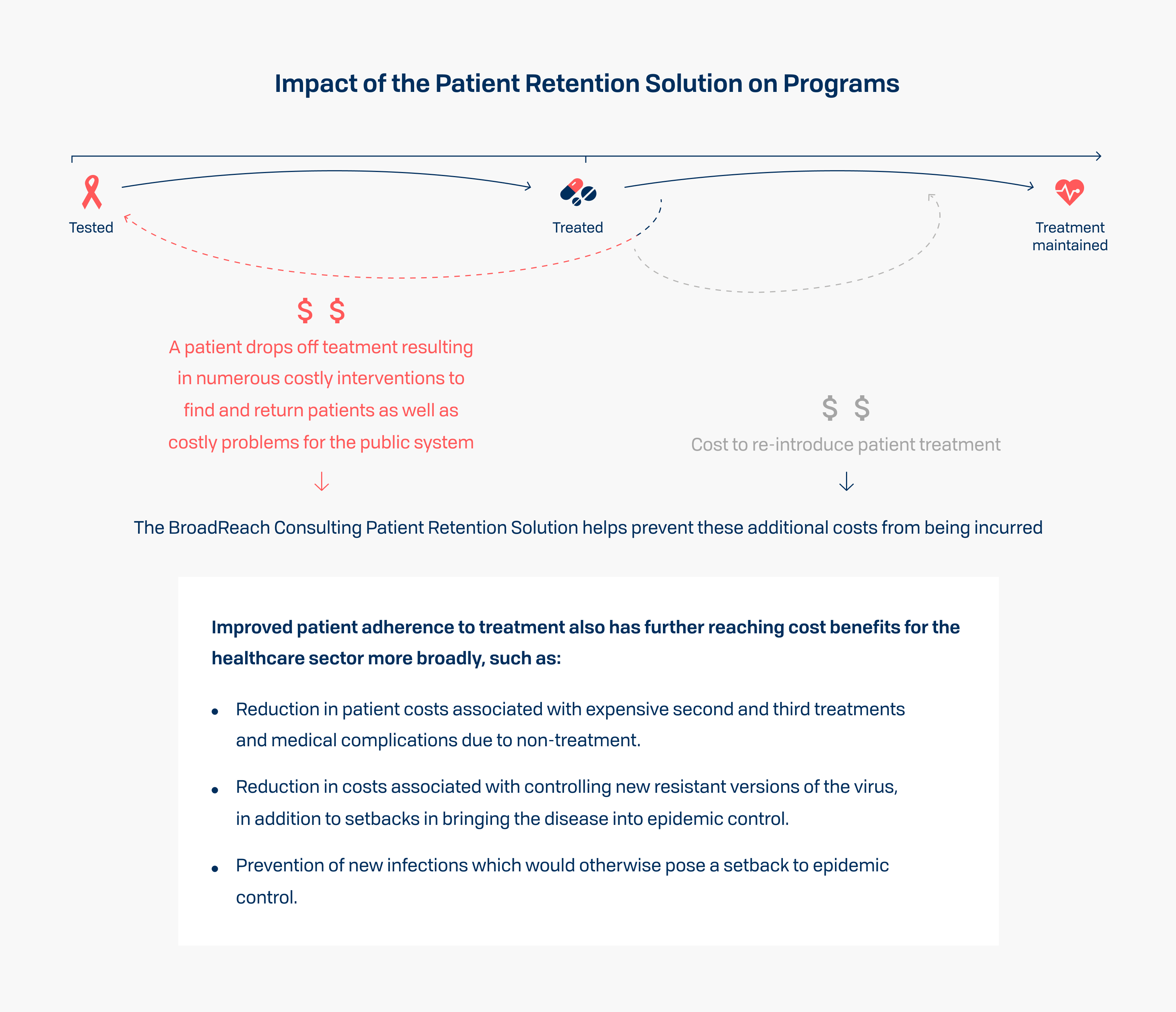
Preventing loss to follow up (LTFU) saves the costs incurred to bring a lost patient back into treatment and avoids catastrophic cost escalation due to issues associated with drug resistance. This ultimately enables programs to spend budgets more efficiently and effectively.
03. Improved efficacy of patient retention interventions
The BroadReach Consulting Patient Retention Solution enables programs to track which interventions yield positive results and which do not, and refine interventions as needed.


Example of an At Risk Patient List produced by the BroadReach Consulting Patient Retention Solution
The power of technology
Folder Number + Gender + Date of Birth (DoB): De-identified data is used to protect patient identity
Date of Birth: DoB is one of many input variables that will influence a patients risk score
Staff Allocated + Cadre of Case Coordinator ‘feature’: Ensuring clear resource commitment to actioning patient retention interventions, the program can measure whether one care giver performs better or worse than another and apply corrective action as well as the customized intervention efficacy
Applied data science
We applied data science techniques to a robust set of HIV electronic register data from more than 300 public healthcare facilities (> 500,000 patients) across districts in two provinces in South Africa.
The goal was to identify factors in the demographic and longitudinal data that were predictive of patients entering a downstream state of the progression towards lost to follow up.
The modeling was designed in accordance with the need for specific predictions aligned to existing clinical workflows, which use predefined heuristics to classify patients according to defined states, in order to target specific interventions relevant for their state.
Predictive analytics that help reduce patient loss to follow up
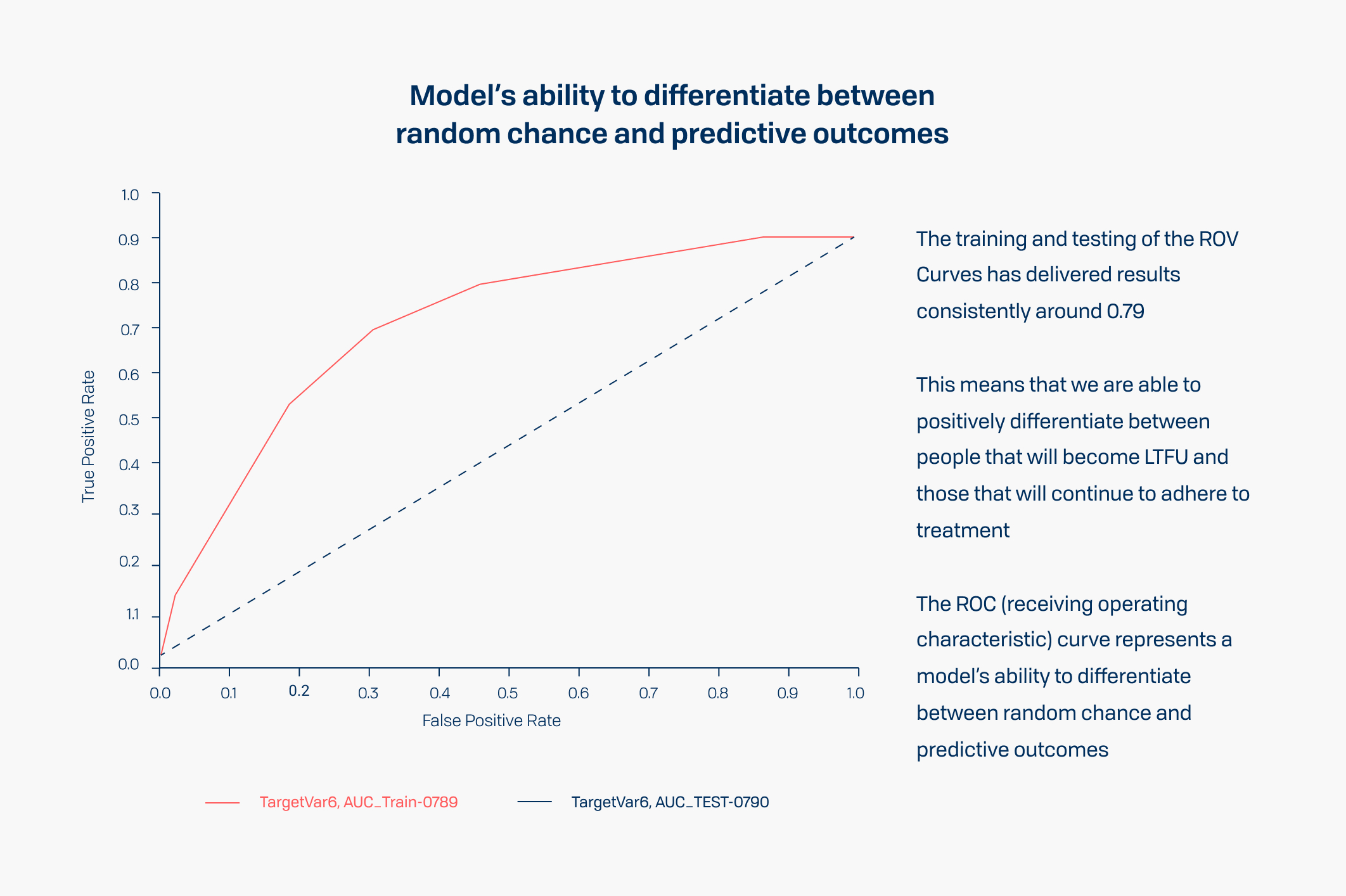
Predictive models were developed using machine learning algorithms to identify the predictive factors, that were then used to generate a risk propensity score.
Our modeling approach considered the need for specificity versus accuracy. In this we were informed not only by the quality of the results, but also by the clinical practice, which in low resource settings is constrained and able only to effectively manage batch processes, rather than personalized patient engagement.
Consistent with good analytics practice, we developed models on a subset of the data, used to train the model, and then applied the resulting models to the broader dataset to validate the quality of the model. We continued to tune the models through numerous iterations of feature and selection.
Example of the common predictive features identified using this process include:
- Season
- Viral Loads at last visit
- Viral load changes over time
- District
- Visit History and missed appointments
- Gender
- Interactions at clinical visit
Our modelling approach was able to achieve sufficiently robust scores across the various predictive models to be insightful at clinical operations level. The confidence in predictions ranged from 68 - 75%, which was then improved to 80 - 88% in the final tuned model.
Models were introduced into a robust processing network, using Vantage Technologies, as a repeatable process to apply the models to near real-time patient data, and to automate direct dissemination of risk propensity scores via email to staff involved in clinical operations on a regular basis.
Contact us
For more information about Vantage Technologies & BroadReach visit www.broadreachcorporation.com